This is relate to our HW 12 last several questions, calculate PH, calculate after adding certain amount of acid and base, the buffer is still effective or not.

Ella Li's List: All my chemistry bookmarks for 109H
-
Dec 04, 08
Nice demonstration about the BRONSTED-LOWRY acid and base definition.
This is relate to the HW 12 Q 1-4
-
Dec 04, 08
HW12 Q18 about buffer, acetic acid/acetate buffer, polyprotic acids
-
equation:


-
Polyprotic Acids
In contrast to a simple monoprotic acid like acetic acid, with only one equilibrium between the acid and conjugate base, a polyprotic acid contains more than one acidic hydrogen. For a polyprotic acid, n acidic hydrogens will exist in solution in equilibrium with n conjugate base forms (for a total of n+1 species). For example, when phosphoric acid (n = 3, a triprotic acid) is dissolved in solution, the following equilibria are established among the four species H3PO4 (phosphoric acid itself), H2PO4-1 (dihydrogen phosphate anion), HPO4-2 (hydrogen phosphate anion), and PO4-3 (phosphate anion):
-
-
Dec 04, 08
This relate to the HW 12 Q 17 which is about how to designing a effective buffer.
-
Dec 04, 08
Buffer's basic theory and formula explanation which would help with HW12 Q12-18
-
Dec 04, 08
This is the explanation of acid-base theory relate to the HW12
-
Dec 04, 08
This is a detail explanation about different acid and base titration process. The good part about this website is it indicate each part of the titration.
-
-
The equivalence point is the place with the most rapid change. Do not confuse with the slowest change place in HW which is the buffer's EP.
-
-
-
this is similar to our lab -two EP
-
-
-
Dec 04, 08
This is a flash tutorial website for the acid-base titration.
I think it's really helpful for our current lab.
The simulator can graph the titration curve as different concentration or volume of Acid and base entered. It's illustrate how the data could be obtained.-
-
Change the numbers of the acid and base concentration and volume. it illustrate the whole titration process and the calculation.
-
-
-
-
Calculate
 H
H ,
, S
S , and
, and G
G for the above reaction to determine whether the reaction is spontaneous or not.
for the above reaction to determine whether the reaction is spontaneous or not. First let's calculate
 Hf
Hf . Note that in the above reaction, one mole of NH4NO3 dissociates in water to give one mole each of NH4+ and NO3-:
. Note that in the above reaction, one mole of NH4NO3 dissociates in water to give one mole each of NH4+ and NO3-: 
Next, let's calculate
 S
S :
: 
-
- Sample free energy calculation

Compound  Hf
Hf
 S
S
NH4NO3(s) -365.56 151.08 NH4+(aq) -132.51 113.4 NO3-(aq) -205.0 146.4 Calculate
 H
H ,
, S
S , and
, and G
G for the above reaction to determine whether the reaction is spontaneous or not.
for the above reaction to determine whether the reaction is spontaneous or not. First let's calculate
 Hf
Hf  . Note that in the above reaction, one mole of NH4NO3 dissociates in water to give one mole each of NH4+ and NO3-:
. Note that in the above reaction, one mole of NH4NO3 dissociates in water to give one mole each of NH4+ and NO3-:
-
-
Nov 14, 08
This spreadsheet will calculate the chemical equilibrium state of an ideal gas mixture, subject to necessary constraints on two intrinsic variables. Depending on the constraint chosen, the calculation invokes STANJAN to minimize the derived property—Gibbs energy, Helmholz energy, internal energy, enthalpy—or maximize entropy for the user-supplied gas mixture.
This relate to our homework assginement and lab. it's really nice for checking answer. -
-
Entropy and probability
As was explained in the preceding lesson, the distribution of thermal energy in a system is characterized by the number of quantized microstates that are accessible (i.e., among which energy can be shared); the more of these there are, the greater the entropy of the system. This is the basis of an alternative definition of entropy
S = k ln Ω (2-2)
in which k is the Boltzmann constant (the gas constant per molecule, 1.38
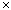 10–23 J K–1) and Ω (omega) is the number of microstates that correspond to a given macrostate of the system. The more such microstates, the greater is the probability of the system being in the corresponding macrostate. For any physically realizable macrostate, the quantity Ω is an unimaginably large number, typically around
10–23 J K–1) and Ω (omega) is the number of microstates that correspond to a given macrostate of the system. The more such microstates, the greater is the probability of the system being in the corresponding macrostate. For any physically realizable macrostate, the quantity Ω is an unimaginably large number, typically around 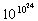 for one mole. By comparison, the number of atoms that make up the earth is about 1050. But even though it is beyond human comprehension to compare numbers that seem to verge on infinity, the thermal energy contained in actual physical systems manages to discover the largest of these quantities with no difficulty at all, quickly settling in to the most probable macrostate for a given set of conditions.
for one mole. By comparison, the number of atoms that make up the earth is about 1050. But even though it is beyond human comprehension to compare numbers that seem to verge on infinity, the thermal energy contained in actual physical systems manages to discover the largest of these quantities with no difficulty at all, quickly settling in to the most probable macrostate for a given set of conditions. The reason S depends on the logarithm of Ω is easy to understand. Suppose we have two systems (containers of gas, say) with S1, Ω1 and S2, Ω2. If we now redefine this as a single system (without actually mixing the two gases), then the entropy of the new system will be S = S1 + S2 but the number of microstates will be the product Ω1Ω2 because for each state of system 1, system 2 can be in any of Ω2 states. Because ln(Ω1Ω2) = ln Ω1 + ln Ω2, the additivity of the entropy is preserved.
-
helpful for the extra lab assignment
-
-
The reason S depends on the logarithm of Ω is easy to understand. Suppose we have two systems (containers of gas, say) with S1, Ω1 and S2, Ω2. If we now redefine this as a single system (without actually mixing the two gases), then the entropy of the new system will be S = S1 + S2 but the number of microstates will be the product Ω1Ω2 because for each state of system 1, system 2 can be in any of Ω2 states. Because ln(Ω1Ω2) = ln Ω1 + ln Ω2, the additivity of the entropy is preserved.
-
-
-
Add Sticky Note
-
A step by step explaination of a typical question.
-
-
-
Nov 13, 08
This is a example of Beer's Law plot for our this week's lab.
-
-
The reaction of aldehydes and ketones with ammonia or 1º-amines forms imine derivatives, also known as Schiff bases, (compounds having a C=N function). This reaction plays an important role in the synthesis of 2º-amines, as discussed earlier. Water is eliminated in the reaction, which is acid-catalyzed and reversible in the same sense as acetal formation.
R2C=O + R'NH2  R'NH–(R2)C–O–H
R'NH–(R2)C–O–H  R2C=NR' + H2O
R2C=NR' + H2O-
the ketone or aldehyde reaction with NH2
-
-
-
-
-
primary alcholol's oxidation form aldehyde
secondary alcholol's oxidation form ketone
-
-
relationship between the primary alcohol and the aldehyde formed.
-
-
-
Carboxylic Acid
R----C===O
.........|
........OH -
Aldehyde
R----C===O
.........|
........H
Ketone
R----C===O
.........|
........R'
-
-
-
Oct 19, 08
this is detail description of biomolecules we learned in class.
It relate to our review exam-the question about the protein secondary structure.-
Add Sticky Note
-
I just curious about the alfa helix interaction. I found this rally nice pic since I thought this might on the exam. This relate to our lab question and it's also relate to our review exam. it;s always NH and C=O interat with each other and from hydrogen bond. There is no interaction occurs between R group or C in the mainchain.
-
-
Secondary structure - this term refers to the arrangement of amino acids that are close together in a chain. Examples of secondary structures are helices and pleated sheets. An alpha helix is a tightly coiled, rodlike structure which has an average of 3.6 amino acids per turn. The helix is stabilized by hydrogen bonding between the backbone carbonyl of one amino acid and the backbone NH of the amino acid four residues away. All main chain amino and carboxyl groups are hydrogen bonded, and the R groups stick out from the structure in a spiral arrangement. As seen in the table below, there are several types of alpha helix that arise from the degree of hydrogen bonding in the helix.
-
-
Oct 19, 08
This is a really nice picture about the side chain interaction between two chains of beta sheet.
This relate to the review exam question #9 and our previous lab.-
Note that the hydrogen bonds holding together the two peptide chains are not 180 degrees
-
Add Sticky Note
-
A question relates to this diagram in the practice exam. The side chain reaction is between O H
-
he interaction is between H from -N-H and O from -C=O
one line is parallel and one line is antiparallel
-
-
List Info
39 items | 14 visits
I collect these bookmarks for homework, lab, and test preperation.
Updated on Dec 05, 08
Created on Dec 05, 08
Category: Schools & Education
URL:

one line is parallel and one line is antiparallel - Ella Li on 2008-10-19